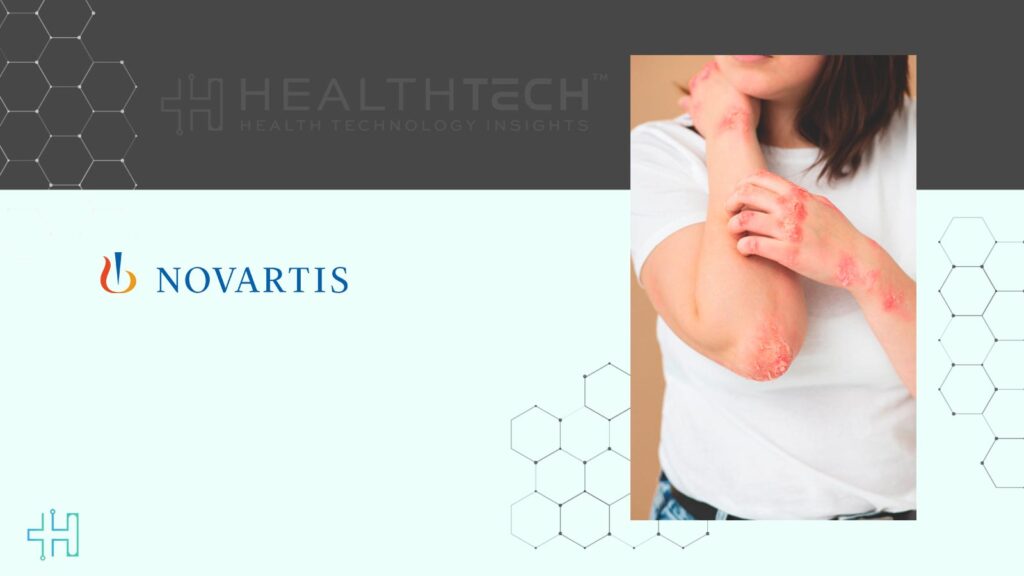
Analyses from REMIX-1 & -2 studies on CSU disease control and early symptom relief will be presented
Novartis will present new data on Rhapsido, a highly selective oral Bruton’s tyrosine kinase inhibitor, at the 2026 American Academy of Allergy, Asthma & Immunology Annual Meeting in Philadelphia, Pennsylvania, taking place from February 27 to March 2. The presentations will include findings from five key abstracts examining Rhapsido’s effectiveness in chronic spontaneous urticaria and its potential as a treatment for peanut allergy, reflecting Novartis’ ongoing commitment to developing therapies for immune-mediated conditions with significant unmet needs.
Health Technology Insights: Corus Orthodontists Secures C$20 Million Equity Investment
Data from the Phase III REMIX-1 and REMIX-2 trials will provide updated insights on disease control and early symptom relief in patients with chronic spontaneous urticaria. Additionally, Phase II results for IgE‑mediated peanut allergy will be presented for the first time in an oral session, highlighting the therapeutic potential of remibrutinib beyond urticaria. Novartis plans to initiate a Phase III clinical program in peanut allergy later in 2026.
Angelika Jahreis, Global Head of Immunology Development at Novartis, emphasized that the peanut allergy findings further support Rhapsido as a well-tolerated oral option for patients with allergic and immune-mediated disorders. She noted that the recent FDA approval for chronic spontaneous urticaria demonstrates the drug’s therapeutic promise and underscores Novartis’ focus on expanding treatment options for patients with few alternatives. Jahreis highlighted that Novartis aims to bring innovative medicines to high-need disease areas to transform patient care and outcomes.
Regulatory reviews for remibrutinib in chronic spontaneous urticaria are ongoing in multiple regions, including the European Union and Japan. In addition to chronic spontaneous urticaria and peanut allergy, remibrutinib is under clinical investigation for chronic inducible urticaria and hidradenitis suppurativa. A submission to the FDA for symptomatic dermographism, the most common type of chronic inducible urticaria, was completed in the fourth quarter of 2025. Positive topline results from the remaining two arms of the Phase III RemIND trial in chronic inducible urticaria were also announced, with full data sets scheduled for submission to health authorities worldwide and planned presentations at upcoming medical congresses.
Through these efforts, Novartis continues to expand the evidence base for Rhapsido, supporting its potential across multiple allergic and immune-mediated conditions while aiming to address unmet patient needs with innovative, orally administered therapies.
Health Technology Insights: SEQSTER Expands Partnership with Praxis Precision Medicines
To participate in our interviews, please write to our HealthTech Media Room at info@intentamplify.com